ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Brom Aoroplasty for Supravalvar Aortic Stenosis
Brom’s three-patch aortoplasty is indicated for the repair of supravalvar aortic stenosis, which can occur as an isolated defect or as a part of Williams syndrom. This syndrome is characterized by supravalvar aortic stenosis, elfin craniofacial features, peripheral pulmonary artery stenosis, and hypercalcemia.
Patient Selection
Brom’s three-patch aortoplasty is indicated for the repair of supravalvar aortic stenosis, which can occur as an isolated defect or as a part of Williams syndrom [1]. This syndrome is characterized by supravalvar aortic stenosis, elfin craniofacial features, peripheral pulmonary artery stenosis, and hypercalcemia. The underlying cause is a loss of function mutation of the elastin gene on chromosome 7q11.23. These patients will have a systolic cardiac murmur and two-dimensional echocardiography can establish the diagnosis. Cardiac catheterization is indicated to evaluate the status of the coronary arteries and assess the pulmonary arterial tree for related peripheral stenosis.
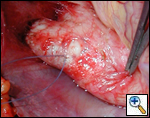
Indications for surgery include symptoms of diminished exercise tolerance, syncope, or angina. Other indications would include 1) systolic ascending aorta gradient greater than 50 mm Hg, 2) aortic valve insufficiency, or 3) evidence of compromised coronary artery perfusion. These patients should undergo surgery at a relatively early age because of the detrimental effect of the high proximal aortic pressure on the coronary arteries with the possibility for accelerated atherosclerotic changes. These patients typically have a thickening of the aortic wall at the sinotubular junction. This abnormal thickening may extend into the ascending aorta and even into the aortic arch. In some patients, this abnormal thickening creates stenosis of the coronary ostium.
The surgical history of operations for supravalvar aortic stenosis is shown in Table 1. Dwight McGoon and John Kirklin [2] from the Mayo Clinic first reported a one-patch teardrop-type repair of supravalvar aortic stenosis in 1961. Donald Doty [3] reported the use of an inverted Y-shaped patch which extended into two of the aortic valve sinuses in 1977. The technique described in this monograph is that of A. Gerard Brom, [4] with three patches placed into the three aortic valve sinuses. This frequently also requires a patch in the distal ascending aorta. Most recently, John Myers [5] described an approach with three incisions into the three coronary sinuses and corresponding counterincisions into the distal ascending aorta which then insert into the openings created in the proximal ascending aorta. This technique has the advantage of not requiring autologous patch material, but is technically more demanding.
Table 1: Surgical History of Operations for Supravalvar Aortic Stenosis
| Surgeon | Technique | Number of Sinuses Opened | Year Reported |
| McGoon | Single-patch | 1 | 1961 |
| Doty | Inverted "Y" Patch | 2 | 1977 |
| Brom | Three-patch | 3 | 1988 |
| Myers | Three-sinus incision | 3 | 1993 |
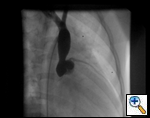
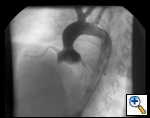
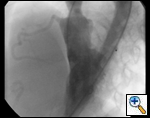
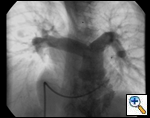
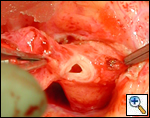
An asymptomatic, 5-year-old, 18-kg boy known by genetic testing to have Williams syndrome on physical examination had a IV/VI harsh systolic ejection murmur and no diastolic murmur. Preoperative echocardiogram showed an aortic root dimension of 2 cm and the dimension at the level of stenosis in the ascending aorta was 8 mm. Peak echocardiogram gradient was 70 mm Hg with a cardiac catheterization gradient of 50 mm Hg. The child had no peripheral pulmonary artery stenosis.
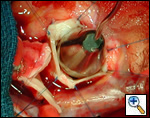
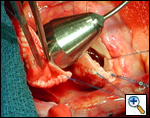
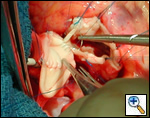
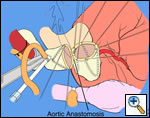
Operative Steps
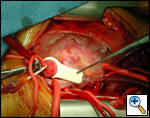
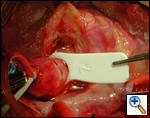
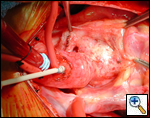
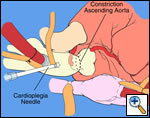
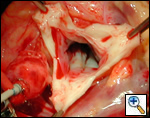
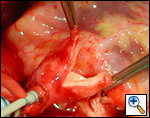
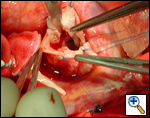
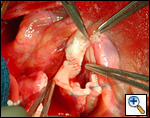
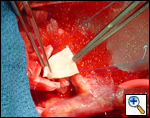
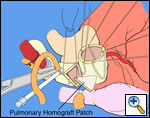
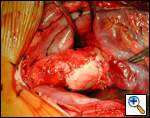
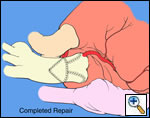
 |
| Figure 1 |
An asymptomatic, 5-year-old, 18-kg boy known by genetic testing to have Williams syndrome on physical examination had a IV/VI harsh systolic ejection murmur and no diastolic murmur. Preoperative echocardiogram showed an aortic root dimension of 2 cm and the dimension at the level of stenosis in the ascending aorta was 8 mm. Peak echocardiogram gradient was 70 mm Hg with a cardiac catheterization gradient of 50 mm Hg. The child had no peripheral pulmonary artery stenosis.
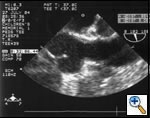
Preoperative transesophageal echocardiogram in long-axis view shows ascending aorta with aortic valve to the left. The markers indicate the aoritc root dimension to be 2.0 cm and hte mid portion of the stenosis to be 0.8 cm.
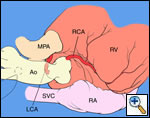
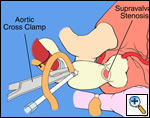
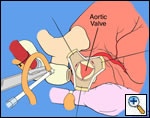
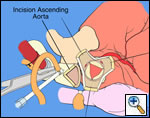
The operative approach is through a median sternotomy with the use of cardiopulmonary bypass and bicaval venous cannulation. The first dose of cold blood cardioplegia is antegrade; the remaining doses are retrograde.
Tips & Pitfalls
-
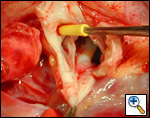
High ascending aortic cannulation adjacent to the innominate artery for maximum exposure of the ascending aorta and to allow length for positioning of the patch in the ascending aorta.
-
One dose of antegrade cardioplegia followed by retrograde cardioplegia.
-
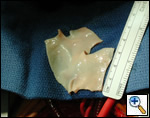
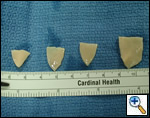
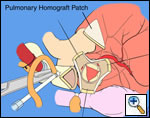
Pulmonary homograft or pericardial patches (tanned in glutaraldehyde) for the “shields”. These patches should not be too large – this will lead to aortic insufficiency.
-
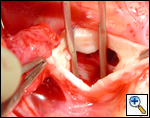
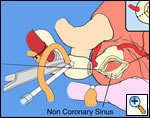
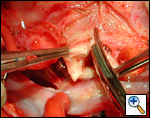
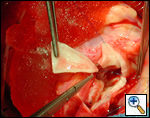
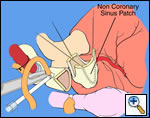
First coronary sinus incision in noncoronary sinus to facilitate exposure of the incisions into the coronary orifice sinuses.
-
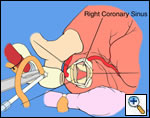
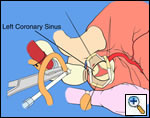
In most cases, the right coronary artery sinus should be opened to the left of the coronary orifice and the left coronary artery sinus to the right of the coronary orifice. Occasionally one may not have enough room between a coronary orifice and the aortic valve; in this case the incision site should be tailored to the situation.
-
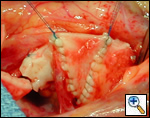
Ensure similar orifice sizes between the proximal and distal ascending aorta after patch augmentation by the use of a sizing dilator.
-
Transesophageal echocardiography to assess completeness of repair, coronary blood flow postoperatively, wall motion abnormalities, and adequacy of aortic valve.
Results
The results of the three-patch aortoplasty technique are shown in Table 2. The report by Stamm and associates from Boston Children’s Hospital, in particular, compared the results of single-patch, bifurcated-patch, and three-sinus reconstruction [7]. Results improved greatly after the introduction of the three-sinus technique, a more symmetric reconstruction of the aortic root. Residual gradients were lower, hemodynamics were superior, and there was a reduction in both mortality rate and need for reoperation.
Table 2. Results of three-sinus incision technique
|
Author |
Year
|
Number of Patients
|
Median Age (Years)
|
% Williams Syndrome
|
Mortality
|
Mean Residual Gradient
|
|
Myers [5] |
1993
|
2 - Brom
4 - Myers |
3.8
|
39
|
0
1 |
12mm Hg
|
|
Hazekamp [6] |
1999
|
13
|
15.8
|
38
|
0
|
10mm Hg
|
|
Stamm [7] |
1999
|
4 - Myers
2 - Brom |
7.4
|
61
|
0
0 |
10mm Hg
|
|
Total |
|
25
|
|
|
1
|
|
References
1. Williams JCP, Barratt-Boyes BG, Lowe JB. Supravalvular aortic stenosis. Circulation 1961;24:1311-8 .
2. McGoon DC, Mankin HT, Vlad P, Kirklin JW. The surgical treatment of supravalvular aortic stenosis. J Thorac Cardiovasc Surg 1961;41:125-33.
3. Doty DB, Polansky DB, Jenson CB. Supravalvular aortic stenosis. Repair by extended aortoplasty. J Thorac Cardiovasc Surg 1977;74:362-71.
4. Brom AG. Obstruction of the left ventricular outflow tract. In Khonsari S, editor. Cardiac surgery: safeguards and pitfalls in operative technique, 1st ed. Rockville, MD: Aspen Publishers, 1988; p 276-80.
5. Myers JL, Waldhausen JA, Cyran SE, Gleason MM, Weber HS, Baylen BG. Results of surgical repair of congenital supravalvular aortic stenosis. J Thorac Cardiovasc Surg 1993;105:281-7.
6. Hazekamp MP, Kappetein AP, Schoof PH, Ottenkamp J, Witsenburg M, Huysmans HA, Bogers AJ. Brom’s three-patch technique for repair of supravalvular aortic stenosis. J Thorac Cardiovasc Surg 1999;118:252-8.
7. Stamm C, Kreutzer C, Zurakowski D, Nollert G, Friehs I, Mayer JE, Jonas RA, del Nido PJ. Forty-one years of surgical experience with congenital supravalvular aortic stenosis. J Thorac Cardiovasc Surg 1999;118:874-85.