ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Completely endoscopic microwave ablation of atrial fibrillation on the beating heart using bilateral thoracoscopy
Patient Selection
Alternative methods for treating atrial fibrillation with surgery have been gaining interest over the last few years. Multiple energy sources are being employed as surgeons seek to create impulse-blocking scars on the right and left atrium. Although most literature reports are small and uncontrolled, it appears that these new technologies are permitting surgeons to perform ablation quickly and safely with promising cure rates in 75 – 80% of patients.
The drive to make ablation procedures more acceptable to a wider group of patients, particularly those with lone atrial fibrillation who otherwise would not be having surgery, has led surgeons to develop minimally invasive methods by which these devices can be introduced into the chest and made to create lesions on the heart. This report describes in detail one such method, the completely endoscopic completely endoscopic microwave ablation of atrial fibrillation on the beating heart using bilateral thoracoscopy.
This procedure was first performed in November of 2002 and reported a few months later. [1] Easy to learn, it has been adopted by many surgeons and, as of this writing, has been performed over 400 times worldwide. Initial reports from several centers confirm that it is safe and appears to be effective at treating patients with AF. [2-7]
The conventional thoracoscopic microwave ablation (MWA) procedure can be broadly divided into five steps, with details given below. In the first step, three access ports are created on the right side of the chest. The pericardial sac is then opened parallel to the phrenic nerve to expose the right superior and inferior pulmonary veins. In the second step, the pericardial reflections investing the superior and inferior vena cavae are dissected to allow entry into the transverse and oblique sinuses, respectively. In the third step, guiding catheters are placed behind the cavae into the sinuses. In the fourth step, three ports are created in the left chest and the left side of the pericardial sac is opened, revealing the guide catheters. In the fifth step, the catheters are retrieved, delivered outside the chest, tied together and then replaced so as to surround the pulmonary veins. In the sixth step, the catheter “loop” is used to draw the Guidant Flex 10 MWA device around the veins. Once the Flex 10’s position is confirmed, a circumferential ablation line is laid down around the pulmonary veins on the left atrial wall. What follows is a detailed description of each step of the procedure, from initial patient workup to postoperative follow-up.
Inclusion criteria:
- Paroxysmal, persistent or permanent atrial fibrillation.
Comment: Although it is more likely that patients with paroxysmal fibrillation will undergo a successful ablation, all patients are considered candidates for the procedure. It is not necessary for the patient to have undergone antiarrhythmic drug trials, cardioversions or attempts at catheter-based ablation. In fact, some will opt for surgery as a first-line therapy. - Negative workup for ischemic coronary disease
Comment: It has been my practice to perform coronary angiography on patients older than 60 years whereas I will accept a negative noninvasive stress test on those younger and without a history of coronary artery disease. This simplifies the patient work-up, as many of these patients are taking coumadin, creating scheduling problems for outpatient workup procedures. - Negative workup for left atrial thrombus and valvular disease
Comment: I will usually accept a negative transthoracic echocardiogram, although it a TEE is frequently required for technical reasons. This is also the opportunity when one should tabulate left atrial dimensions and function. - The remainder of the workup is per routine for your institution, including a chest x-ray and any laboratory data needed prior to the administration of general anesthesia.
Exclusion criteria:
- Previous cardiothoracic surgery
Comment: I consider this to be an absolute contraindication. Previous cardiac surgery, previous thoracic surgery and/or previous pericardial procedures will leave the patient with extensive adhesions, particularly in the transverse sinus where aortic cross clamps have been placed or behind the cavae when bicaval cannulation has been performed. Undertaking an adhesion takedown in these areas through an endoscope is not advised, regardless of the operator’s skill. - History of pleural disease
Comment: Prior pneumonia or pericardial effusion is a relative contraindication, as adhesiolysis of the lung can usually be performed endoscopically. Although it is tedious, the operator is usually rewarded with a normal pericardium and the procedure can continue normally once the pericardium is opened. - Known left atrial thrombus
Comment: The presence of a thrombus in the LA or in the LA appendage is an absolute contraindication. The preoperative discussion with the patient should include this possibility and whether or not the patient wants the surgeon to abandon the procedure or continue with an open, endocardial approach that would likely require sternotomy and cardiopulmonary bypass (although such an approach can certainly be performed through a less-invasive approach, it would still mandate cardioplegic arrest and endocardial exposure). - Advanced pulmonary disease suggesting intolerance of single-lung ventilation.
Comment: This is currently the most difficult quality to assess prior to operation. A history of pulmonary disease and/or tobacco abuse should compel the surgeon to investigate further. Although it is possible to perform the procedure on a patient who does not tolerate single-lung ventilation well, it becomes very tedious and can be dangerous.
Additional Comments
There are no specific exclusions with regard to age, left atrial size or duration of the fibrillation. To date, the procedure has been performed on patients with all forms of AF, ranging in age from 38 to 82 years, on patients with left atria of normal size as well as those with enlarged atria, and on those with long-standing fibrillation of 20 years as well as recent onset fibrillation as short as 6 months. Although the “optimal” patient would have failed multiple attempts at drug therapy, have had least one attempt at catheter-based ablation, be experiencing highly symptomatic paroxysmal fibrillation and have a small atrium, this is not strictly necessary and the delineation of further inclusion and exclusion criteria will await time and experience.
Preoperative preparation
A detailed discussion should take place between the surgeon and the patient with regard to the procedure, its expected outcome and possible complications. Contingency plans should be discussed in the case that extreme technical difficulties are encountered and the surgeon cannot proceed endoscopically. Should a sternotomy be performed? Should a thoracotomy be performed? Should the procedure be abandoned?
Operative Steps
Patient positioning and preparation
The patient is brought to the operating room and placed on the operating table in the supine position, where double lumen general endotracheal anesthesia is induced. The use of bronchial blockers or UniVent® tubes is to be discouraged, as complete control over both lungs individually is mandatory.
A transesophageal echocardiogram probe is inserted and the left atrium sized and inspected for thrombus in the appendage. As discussed above, if thrombus is found the procedure should be abandoned or converted to an open, on-pump endocardial ablation with clot extraction. The probe is sometimes withdrawn after assessment in order to help with passage of the guide catheters, but this is not absolutely necessary. A large bore venous access line is then secured, either through the antecubital veins or the internal jugular vein. Radial arterial access is obtained for blood pressure and arterial blood gas monitoring. A pulmonary artery catheter is not necessary and will actually narrow the bore of the sheath through which it is placed, making large volume resuscitation more difficult if needed.
The arms are abducted at 90 degrees and placed on arm boards. A small rolled towel or sheet is placed vertically behind the spine to thrust the chest slightly forward and further extend the arms. The axillae and chest are shaved and a sterile prep is performed from the chin to the knees and from the posterior axillary line on one side to the other.
Initial pericardial entry and exposure
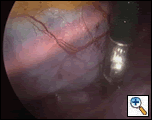
The right lung is then deflated and the right pleural space entered with a 5mm valved access port in the 4th intercostal space along the anterior axillary line. A valved port is used so that CO2 can be insufflated at ~10mmHg pressure to rapidly create a pneumothorax. Some surgeons are using open “ThoracoPorts” but I have found that this makes moving from the right to the left side more time consuming. A 5mm 0-degree telescope is inserted through the port and the intrapleural contents inspected, with particular regard to the angle of view toward the pericardium (Figures 1 and 2).
 |
 |
| Figure 1: Planned right pericardiotomy | Figure 2: View through right-sided port |
Two additional working ports are created, one in the 3rd intercostal space along the anterior axillary line (5mm) and another in the 5th intercostal space along the midclavicular line (10-12mm). The ports should be placed so that the surgeon does to “fight” with the assistant and so that the angle of attack to the pericardium is as oblique as possible.
The phrenic neurovascular bundle is then visualized through the endoscope. The pericardial sac is opened using endoscopic graspers and shears along a line parallel to and 2 cm anterior to the phrenic nerve bundle. The incision begins where convenient and is extended superiorly until the aorta is visualized. Typically the grasper would be placed in the left hand and the shears in the right. Once the incision is carried far enough superiorly, the instruments are swapped and the incision extended inferiorly down to the diaphragmatic fat pad. It is not necessary to incise into the fat pad as this just results in troublesome bleeding and no real benefit for exposure.
Comments
One of the most difficult things to learn is when the port locations are correct to perform the procedure. If visualization is a problem, then a 30° angled 5mm telescope may solve this problem. If the endoscopic instruments cannot be manipulated into the correct positions, then new 5mm ports should be created as needed. The resulting cosmetic insult from a 5mm port is minimal and procedural safety and comfort should not be sacrificed for the creation of one or two extra ports.
Once the pericardium is opened, it is very rarely necessary to place any kind of fixed retraction on it. The bulging of the right atrium will spread the pericardiotomy widely and permit visualization of the atrial free wall, aorta and vena cavae. The pulmonary veins themselves are harder to see, and their exposure is described in detail below.
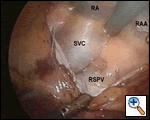 |
| Figure 3: Right-sided port view, showing superior vena cava (SVC), right superior pulmonary vein (RSPV) and right atrium (RA). Dissection begins posterior to SVC and medial to RSPV. |
Pericardial reflection dissection into the transverse and oblique sinuses
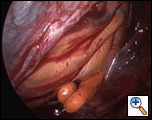
Access to the transverse and oblique sinuses is obtained by dissecting away the pericardial reflections behind the superior and inferior vena cavae, respectively. This can be done in either order, although I usually begin dissection behind the superior cava, just cephalad to the right superior pulmonary vein (Figure 3).
Grasping the cut pericardial edge, avoiding the phrenic nerve, and placing it on traction rightward toward the operator achieves exposure to the pulmonary veins. One then places a blunt 5mm suction instrument into the angle formed by the SVC and the RSPV. (The use of a suction instrument, rather than a formal dissecting instrument, allows one to remove any blood or fluid that may accumulate in this space.) The surgeon then gently “lifts” the SVC anteriorly while exerting very gentle pressure inward and leftward toward but under the aorta. This results in a controlled tearing of the pericardial membrane after a few strokes. If necessary the telescope can actually be advanced quite far into this space while holding up on the SVC in order to confirm that the dissection has been adequately completed. When completed, the telescope can often be driven beneath the SVC and into the transverse sinus with no resistance felt. As a general point, when the surgeon is first learning this technique, he or she should move slowly and be patient until “the feel” of the dissection has been learned.
Once the transverse sinus has been dissected, attention is turned to the oblique sinus. After placing the blunt dissector into the angle formed between the RIPV and IVC, the surgeon gently lifts the IVC anteriorly while pressing inward and leftward. This pericardial reflection is much thinner and more easily breached than that behind the SVC, and the oblique sinus is usually easily entered. One can easily confirm this by directly visualizing the posterior wall of the left ventricle beneath the IVC.
Comments
It must be emphasized that the motions should be gentle yet persistent and under direct visualization. Although some surgeons have used robotic techniques, I consider the loss of tactile feedback to be a significant detraction to this method. Occasionally, a patient may present with an elevated right hemi-diaphragm, occluding one’s view of the oblique sinus approach. By extending the inferior-most port on the right side one can introduce an “EndoFan” or similar instrument and use it to displace the diaphragm inferiorly, enhancing exposure. Other surgeons have described using a suture on the dome of the diaphragm and placing it on traction laterally, drawing the diaphragm inferiorly and clearing the field. I have generally not found this to be necessary but may prove useful from time to time.
Placing guide catheters in the sinuses
As the Flex 10 MWA probe cannot be steered or directed, it is necessary to substitute a guidance mechanism that can be used to place the device. This is done by inserting red Robinson catheters beneath each of the cavae and through the sinuses to the left side. The catheters are stiff enough to allow straight passage through the sinuses yet soft enough not to perforate any structures if they are used with gentle pressure.
One catheter is used for each sinus. Any size is acceptable, and I have used catheters measuring between 14 French and 18 French. If red Robinson (Robnel®) catheters are not available, the clever surgeon can substitute other similar devices although I would suggest avoiding clear tubes, as these are difficult to see through the endoscope. Just prior to placement, the catheter is stiffened and made more “directable” by inserting a slightly curved, lubricated urological stylet in its lumen. The lowermost (10 – 12mm) trochar is then and the catheter is then introduced through the incision. It is then steered into the sinus by going under the SVC or IVC, respectively, under direct vision. (Trochar removal enables the surgeon to place the catheter as far as possible into the chest and across to the left side. It also protects against inadvertent catheter dislodgement by pulling out the trochar and catching the end-flare of the Robnel on the trochar valve mechanism.)
It is important to note that the stylet is not passed any further into the sinus than just beneath the caval vein. Once the tip of the probe has passed beneath the vein, the stylet is held stationary and the catheter is pushed off the stylet into the sinus. When completely inserted, the flared end of the catheter is clipped to the port site with a small hemostat to prevent loss into the pleural space. Because this maneuver eventually results in two flared ends being clipped to the skin at the same location, the surgeon should develop a system by which he or she can keep track of the catheters. (My alliterative mnemonic: Straight hemostat for superior vena cava catheter.)
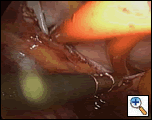 |
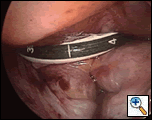
| Figure 4: Videoscopic view from the right pleural space of both guide catheters in place. |
Once the stylets are correctly positioned, the instruments are removed from the right pleural space and the right lung reinflated. Figure 4 depicts the guide catheters in place beneath the caval veins, going through the transverse and oblique sinuses.
It is very important to “feel” the catheters travel under the caval veins and into the sinuses. There can be no hindrance to passage, as this would indicate malpositioning of the device; the catheter must pass easily and without resistance. If resistance is encountered, it is recommended that the suspect catheter be removed, the blunt dissector be replaced into the chest and used to lift the vein while the camera is placed beneath the vein and the dissection inspected. This is most likely to occur beneath the SVC as it is the more difficult dissection; the oblique sinus dissection is usually quite easy and direct visualization of the left ventricle clearly confirms that there is no false plane or passage. If necessary, additional dissection can be carried out to achieve full entry into the sinus(es). (Hint: Dissection and placement of the transverse sinus catheter is usually easier if the transesophageal echo probe is drawn back or removed.)
Sometimes it is necessary to withdraw and reinsert the guide catheters while positioning them. The surgeon is advised to be extremely careful then reinserting the stylet into the catheters, as the stylet often protrudes through the side hole of the catheter, making the assembly into a weapon for perforations. It is therefore recommended that should the stylet become dislodged or withdrawn from the catheter for any reason both the stylet and catheter be removed from the chest, reassembled outside the chest and then the assembly reinserted for a fresh attempt at instrumentation of the sinuses.
Left side pericardiotomy, retrieval of the guide catheters and placement of the MWA device
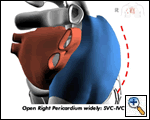
The right lung is now reinflated and the left lung deflated. The left chest is entered in the same manner as the right, with a “mirror-image” port pattern (Figure 5). The only exception to that is that the inferior-most port should be positioned along the posterior axillary line in order to optimize the approach angle to the base of the LA appendage. Again, the use of valved ports and low-pressure CO2 insufflation will rapidly produce a pneumothorax.
The pericardium and phrenic nerve pedicle are visualized, as in Figure 6. It is not uncommon at this point to see one or both of the guide catheters through the pericardium, and their locations should be noted. As the left phrenic nerve rides much more anteriorly on the pericardium than the right, the left side of the pericardium is entered this time posteriorly to the pedicle. A 3 – 4cm pericardiotomy is performed using the scissors and graspers, about 1cm anterior to the pericardial reflection with the hilum of the lung. It is not uncommon to encounter a gush of serosanguinous fluid upon opening but no frank blood should be seen.
At this point it is common to see the transverse sinus guide catheter exiting above the left superior pulmonary vein (LSPV). It may be easily retrieved and delivered out the inferior-most port on the left side. Occasionally, one may find that the guide catheter is not long enough to make this traversal, particularly if the patient is large or barrel-chested. By suturing another rubber catheter to the flared end of the transverse sinus catheter (on the right side), one can effectively “elongate” this tube enough to satisfactorily deliver it outside the chest wall on the left side.
Retrieval of the oblique sinus catheter can be more challenging, particularly if the tip of the catheter has fallen toward the apex of the heart rather than superiorly toward the left inferior pulmonary vein (LIPV). If the oblique sinus catheter is not seen immediately, usually an extension of the pericardiotomy inferiorly combined with a gentle lifting of the ventricle anteriorly will expose the catheter. It can then be purchased with a grasper and likewise exteriorized through the inferior-most port.
Once the tips of the two catheters are available extracorporeally to the surgeon, they are sewn together with a simple stitch of 2-0 or 3-0 silk. Traction is then placed on the flared ends of the catheters from the right side of the chest and the whole assembly is seen to come to rest posterior to the left atrial appendage and around the pulmonary veins. It is very important to observe this step directly (Figure 7), as malposition usually results in a guidance system wrongly located anterior to the LA appendage with resultant ablation of the circumflex coronary artery.
The Flex 10 MWA probe is then sutured to the flared end of the transverse sinus catheter. Traction is placed on the oblique sinus catheter and the Flex 10 is introduced into the right chest while observing its progress from the camera still positioned in the left pleural space. It is important to thread the Flex 10 into the right chest with the flat surface of the probe (the antenna side) facing inferiorly, so that the antenna will come to rest against the dome of the left atrium through the transverse sinus. If the probe is seen to be coming around the left pulmonary veins in an incorrect orientation, the handle of the device may be rotated and the threading process proceeds and usually this will correct the problem. Always confirm that the Flex 10 remains posterior to the LA appendage as it is threaded (Figure 8).
The Flex 10 is threaded far enough into the chest so that position #4 or #5 is seen coming to rest between the LSPV and the LIPV, snugly seated in the angle between the veins and the left atrium. Once this position is confirmed, the thoracoscopic instruments are removed from the left chest and the left lung reinflated.
The right lung is deflated and a camera introduced into the right pleural space. The antenna portion of the Flex 10 exiting from the transverse sinus beneath the SVC is confirmed to be resting correctly against the dome of the atrium; rotation is performed as needed. The antenna portion of the probe exiting from the oblique sinus is likewise noted to be resting against the floor of the left atrium as it exits beneath the IVC. The rule to remember is “black line away from the heart, antenna toward the heart.”
N.B.: It is important to realize that the plane of the antenna should not be constant as it encircles the left atrium. The plane begins vertically upon entering the transverse sinus, twists to 45° as it wraps around the left-sided pulmonary veins and finishes almost horizontally as it exits the oblique sinus. Attempts to force the plane into one orientation will result in failure of contact between the device and the atrium, mainly between the lower pulmonary veins, resulting in poor myocardial penetration.
Ablation of the atrium
The first ablation site is determined by inspecting the markings on the shield side of the Flex 10 as it exits the oblique sinus. This is usually location #1 or #2. The antenna is positioned appropriately as marked on the Flex 10 handle and the ablation is performed with a power setting of 65 watts for 90 seconds per lesion. Gentle traction should be maintained continuously on the Flex 10 as it exits the right-sided port in order to prevent any gaps from forming as the heartbeat and respiratory cycle will tend to pull the device into the chest and loosen it. Once an ablation is completed, the handle is moved to the next location and the next site ablated, as shown in Figure 9. The ablation is complete when the antenna has been positioned at the final site exiting beneath the SVC and activated.
Because the ablation sequence has taken place with the Flex 10 in a “horseshoe” configuration around the veins, there will be a gap of unablated tissue between the RSPV and the RIPV. Grasping the Flex 10 with the endoscopic grasper and pressing it inferiorly toward its origin under the IVC closes this gap (Figure 10). The antenna is positioned appropriately as indicated by the marking on the shield and an ablation line laid down, usually requiring two or three activations. Closure of the gap is confirmed by visual inspection.
In order to prevent left atrial flutter from appearing after the procedure, a lesion is placed from the left atrial appendage into the transverse sinus, connecting to the pulmonary vein “box” lesion. This is done by removing the endoscopic instruments from the right chest, reinflating the right lung, deflating the left lung and reestablishing access to the left pleural space. The Flex 10 is removed from the sulcus around the left pulmonary veins by grasping it with an endoscopic grasper, placing it on traction and lifting it anteriorly toward the sternum, as
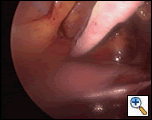 |
| Figure 11: Creating an ablation line onto the LA appendage. The Flex 10 is held upward toward the sternum against the appendage while two or three lesions are created. |
shown in Figure 11. The antenna side of the device is held against the appendage while the device is activated to create a lesion. This usually required two applications to complete.
Amputation of the appendage
The Flex 10 may now be withdrawn from the chest by placing traction on the handle of the device and observing as it exits the chest. An endoscopic stapler (EndoGIA, EndoPath or Power Medical SurgASSIST) is placed through the inferior-most left-sided port and guided to the base of the LA appendage. It is important to manipulate the appendage itself as little as possible to avoid tearing and bleeding. By gently manipulating the stapler and pressing it against the atrium it is possible to capture the appendage. The stapler is then closed carefully, using TEE to confirm that the appendage orifice has been occluded. A single application of the stapler is usually sufficient to complete the closure and amputation.
The specimen is then removed from the chest and the staple line inspected for bleeding, which in my experience is quite rare. In the unusual event that there is bleeding, it will usually stop after application of pressure with an EndoPeanut or similar device for a few minutes. Occasionally, a large HemoClip can be used to complete hemostasis.
Comments
The use of the computer-operated stapler has made the amputation procedure safer and more reliable, with less manipulation of the appendage. Although I do not usually use reinforcing pericardial strips, some surgeons have found this quite useful and it is recommended. In the event that bleeding is encountered from the appendage, a small left thoracotomy should be performed expeditiously and the bleeding controlled manually; control with endoscopic instrumentation is to be discouraged as the bleeding can be quite brisk.
Completion
A 28 French chest tube is placed in each pleural space and placed on routine PleurEvac suction. Full ventilation is resumed. Liberal amounts of 0.25% bupivicaine are infiltrated locally around all of the port sites. The wounds are closed in the usual fashion. The patient is awoken and extubated in the operating room. At UMass we observe these patients in the ICU for the night, but this is probably not necessary.
Results
Post-operative management
Patients can usually have their chest tubes removed in a few hours, after they are stable and a chest x-ray shows no residual pneumothorax or effusion. They are started back on their usual dose of coumadin and any antiarrhythmic drug they were taking prior to surgery. Oral pain medication is given as needed.
Patients can usually be discharged the next morning. They are followed in the office in about one week, at which time a chest x-ray is obtained and they are examined. They are seen thereafter at the 1-, 3-, 6-, 9- and 12-month anniversaries of their procedure at which time an EKG and an echocardiogram are performed. Atrial fibrillation is extremely common after this procedure, occurring in 56% of my patients, and so they are maintained on anticoagulant and antiarrhythmic therapy for at least three months wherein these medications may be stopped if they show stable sinus rhythm.
If patients remain in atrial fibrillation after three months’ time, I will arrange for an outpatient DC cardioversion. Several patients have achieved and remained in sinus rhythm after this approach. If they continue to exhibit stable sinus rhythm for another three months, I will discontinue their medications.
References
- Saltman AE, Rosenthal LS, Francalancia NA, Lahey SJ. A completely endoscopic approach to microwave ablation for atrial fibrillation. Heart Surg Forum. 2003;6:E38-41.
- Salenger R, Lahey SJ, Saltman AE. The completely endoscopic treatment of atrial fibrillation: report on the first 14 patients with early results. Heart Surg Forum. 2004;7:E555-8.
- Reade CC, Johnson JO, Bolotin G, Freund WLJ, Jenkins NL, Bower CE, Masroor S, Kypson AP, Nifong LW, Chitwood WRJ. Combining robotic mitral valve repair and microwave atrial fibrillation ablation: techniques and initial results. Ann Thorac Surg. 2005;79:480-484.
- Pruitt C. Personal communication. STS Annual Scientific Sessions. 2005
- Loulmet DF, Patel NC, Patel NU, Frumkin WI, Santoni-Rugiu F, Langan MN, Subramanian VA. First robotic endoscopic epicardial isolation of the pulmonary veins with microwave energy in a patient in chronic atrial fibrillation. Ann Thorac Surg. 2004;78:e24-5.
- Jansens JL, Ducart A, Preumont N, Jottrand M, Stefanidis C, Stoupel E, de Canniere D. Pulmonary vein isolation by robotic-enhanced thoracoscopy for symptomatic paroxysmal atrial fibrillation. Heart Surg Forum. 2004;7:E595-8.
- Garrido MJ, Williams M, Argenziano M. Minimally invasive surgery for atrial fibrillation: toward a totally endoscopic, beating heart approach. J Card Surg. 2004;19:216-220.