ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Endovascular Repair of Abdominal Aortic Aneurysm (EVAR)
Kruse M, Khoynezhad A. Endovascular Repair of Abdominal Aortic Aneurysm (EVAR). November 2022. doi:10.25373/ctsnet.21526554
Patient Selection
Indications for endovascular repair of the abdominal aortic aneurysm are:
- Patients with asymptomatic abdominal aortic aneurysm greater than 5 cm in an orthogonal projection or larger than twice the size of the normal abdominal aorta or an aneurysm diameter of 4–5 cm, which has also increased in size by 0.5 cm in last six months; there must be no evidence of connective tissue disorder.
-
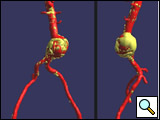
Symptomatic aneurysms mandate endovascular (or open) repair regardless of size. The patient should have a proximal landing zone (distal to renal arteries) of at least 1.5 cm without significant calcification or thrombus, and similarly a distal landing zone of at least 2.5 cm. The proximal neck angulation should be less than 45 degrees, and the patient should have access vessels of at least 6-8 mm luminal diameter (depending on the manufacturer’s requirements) and without extreme tortuosity (Figure 1). Depending on device availability, the patient’s landing zone diameter has to be within the manufacturer’s instruction for use (IFU), for example the Medtronic AneuRx AAAdvantage device is indicated for an infrarenal aortic neck diameter of 16-25 mm (10-20% smaller than labeled device) and iliac diameter of 10-18 mm. Individual consideration should be given for patients who may display some, but not all, of the above indications.
Operative Steps
- All endografts currently FDA-approved for AAA repair are modular systems, except for the Endologix Powerlink, which has a unibody, bifurcated design. Each endograft system has a distinct deployment mechanism, which makes it critical to be completely familiar with the manufacturer’s IFU when planning the procedure.
- The patient is placed in the supine position. Both groins and the entire abdomen are prepped and draped. EVAR may be performed under general, regional or local anesthesia.
- Bilateral access to the common femoral artery is obtained via femoral artery cutdown. Alternatively, in some cases EVAR can be performed percutaneously. The closure device is introduced prior to inserting large sheaths or the endograft device (“preclose technique”). The left brachial/radial artery is accessed only if needed for the body floss maneuver or if there is difficulty cannulating the contralateral limb gate of the endograft.
- The patient is heparinized for a target activated clotting time ≥ 300 sec. This level of anticoagulation is maintained throughout the procedure, until the femoral artery cutdown is repaired and distal pulses are verified. Bilateral access starter guidewires are advanced under fluoroscopic guidance across the lesion in the abdominal aorta.
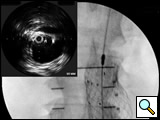
- Initially a 9F introducer sheath is inserted over the primary access guidewire in the more agreeable femoral artery. If there is a concern about aortic rupture, a 12F sheath may be used in order to accommodate large diameter occlusion balloon. A 5F Bern or multipurpose catheter is introduced to facilitate guidewire exchange to a stiff wire (such as the Meier wire or Amplatz). This wire will straighten tortuosity of the access vessel and improve tracking capability of the introduced catheters and devices. An intravascular ultrasound (IVUS) catheter is advanced over the stiff guidewire for inspection of the abdominal aorta. The use of IVUS allows the surgeon to interrogate the entire abdominal aorta and the iliac vessels and to map out (on the fluoroscopic screen) the renal and internal iliac arteries without the use of contrast/fluoroscopy. On the contralateral side, a 5F pigtail catheter to be used for angiography is introduced over the initial guidewire.
- The pigtail catheter is used to perform an aortogram of the abdominal aorta and the iliac arteries. After the angiogram is performed, the proximal neck is evaluated. The length and the diameter of the proximal and distal neck are measured using the preoperative CT scan and the intraoperative IVUS, as well as the angiogram. The details of these imaging modalities will be discussed within the Endoluminal Technology Portal. Based on these measurements, the main trunk’s and iliac arteries’ length and diameter are chosen. On the side of the device’s main trunk and ipsilateral limb introduction an 18-24F sheath (depending on manufacturer’s requirements and size of the stent graft) will eventually be inserted, and on the contralateral side a 12-17F sheath.
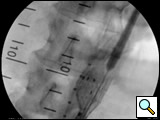
- The main trunk is flushed with heparinized solution, advanced into the proximal neck, positioned just inferior to the lowest renal artery, and oriented so that the contralateral limb gate can be easily accessed. The orientation of the contralateral gate is performed using the manufacturer’s radiopaque markers at the crown of the trunk and/or the stent graft bifurcation under fluoroscopic guidance. A repeat angiogram is commonly performed to reconfirm the positioning of the device within the aorta. Subsequently, the main trunk is deployed (Figures 2, 3), opening the contralateral limb gate.
- The contralateral limb of the endograft will be introduced over another stiff guidewire inserted from the contralateral femoral artery. Deployment of the contralateral limb of the endograft is most often accomplished through retrograde cannulation of the contralateral limb gate of the main trunk, but other methods can be employed if difficulty is encountered. Cannulation of the contralateral limb gate from a retrograde femoral artery approach is commonly achieved by first using a maneuverable guidewire and steerable catheter. Antegrade or crossover cannulation involves passing a guidewire from the ipsilateral limb to the contralateral limb gate of the endograft, which can be accomplished with a curved catheter (such as SOS-Omni). The wire may be retrieved on the contralateral limb using a snare device. Cannulation can also be achieved using an approach from the brachial/radial artery. To confirm that the guidewire is inside the endograft and not between the graft and vessel wall, a pigtail or other curved catheter can be inserted over the guidewire. The ability to rotate the catheter freely without resistance provides a “relative” assurance that the guidewire is within the endograft (rather than between the endograft and the aortic wall). Also, anteroposterior and oblique fluoroscopic views may help confirm the location of the wire. However, while these “tricks” work well in a majority of cases, they may fool the surgeon in some instances. The only accurate way to confirm the location of the wire within the contralateral gate is to advance the IVUS catheter and visualize the contralateral gate.
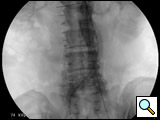
Figure 5: The stent graft is completely deployed and ballooned. Angiogram reveals patent bilateral renal and internal iliac arteries.
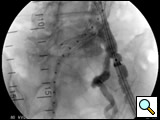
Figure 4: Deployment of contralateral limb. Angiogram reveals the location of the internal iliac artery.
- After the deployment of the contralateral iliac limb (Figure 4), the surgeon will evaluate the need for further iliac limb extenders or aortic cuffs. It is important to cover the entire length between the lowest renal artery and the internal iliac artery. The stent grafts are subsequently ballooned as suggested by the IFU. An IVUS interrogation of the entire stent graft and surrounding aortic branches is performed. This will detect any circumferential stent mal-apposition to the proximal or distal landing zone that may lead to endoleak. A completion angiogram is performed to confirm lack of gross endoleak (Figure 5, Video 1, , “Completion angiogram” confirms the patency of both renal arteries and lack of proximal endoleak (type Ia).) However a single aortogram may miss endoleak due to projection overlap. A bi-plane aortogram is more reliable in excluding any significant procedural endoleak.
- After the completion angiogram, the introducer sheath is removed while carefully leaving the wire in the aorta. If there is any concern about iliac/femoral artery injury, an angiogram is performed. If no angiogram is performed, and the patient remains hemodynamically stable, an intact iliac/femoral artery may be assumed. In this case, the wire is removed, and the femoral artery is repaired in standard fashion. Alternatively, the “perclose” technique may be used to close the arteriotomy.
- The distal pulses are checked. If they correspond to preoperative baseline, the heparin is reversed. At this time, the sterile endovascular wires/catheters may be disposed.
Preference Card
Sheaths
- 9F sheath (Terumo 9fr x 10cm Pinnacle Sheath) is used for initial femoral artery access.
- 6F sheath (Terumo 6fr x 10cm Pinnacle Sheath) used for initial percutaneous access of femoral artery.
- 4F sheath (Terumo 4fr x 10cm Pinnacle Sheath) used for left brachial/radial artery if needed for body floss maneuver or contralateral limb gate cannulation.
- Micro-Puncture Introducer Set (Cook, Inc. 4fr, MPIS-401).
- 18-24F sheath as per stent graft manufacturer’s IFU.
- Large 12 or 14F sheaths (Terumo Pinnacle or Cook Check Flo) should be available for post-deployment molding of the graft with large-diameter occlusion balloons.
Catheters
- 5F Bern (Boston Scientific 5fr x 100cm Imager II Selective- Berenstein tip) or angled tip catheter used for ipsilateral aortic access.
- 5F pigtail catheter (Cook, Inc. 5fr x 90cm Pigtail-Royal Flush Plus) used in the contralateral femoral artery.
- 4F Bern (Boston Scientific 4fr x 100cm Imager II Selective) or angled tip catheter (if brachial/radial artery access is used).
- 8.2F IVUS catheter (Volcano, 8.2F x 90cm Vision PV, 8.2F, 8-12 MHz).
Guidewires
- 0.035 x 180cm Bentson Starter guidewire (Boston Scientific 0.035 x 180cm Starter).
- 0.035 x 180cm hydrophilic guidewire (Terumo 0.035 x 180cm Glidewire).
- 0.035 (or 0.025) x 260cm Stiff type guidewire with soft tip (Boston Scientific 0.025 x 260cm Platinum Plus ST Guidewire; Boston Scientific 0.035 x 260cm Amplatz Super Stiff Guidewire, 6 cm tip length; Boston Scientific 0.035 x 260cm Meier Guidewire, 10 cm tip length; Cook, Inc. 0.035 x 260cm TFE Coated Lunderquist Guidewire, 15 cm tip ; ev3, Inc. 0.035 x 260cm Nitrex wire).
- 0.035 x 450cm hydrophilic guidewire (Terumo 0.035 x 450cm Glidewire).
Balloons
- 8mm x 8cm angioplasty balloon (Boston Scientific 8mm x 8cm Ultrathin Diamond Balloon).
- Large diameter (~ 40mm) compliant occlusion balloons (Medtronic 12fr Reliant Stent Graft.
- Balloon, 46mm max diameter; Cook, Inc., 14fr CODA Balloon Catheter, 40mm max diameter).
Miscellaneous
- Dilator (Endovascular Dilator Set, Cook, Inc. 20-24 fr).
- Snare (Medical Device Technologies 27-45mm EN Snare).
- Luer-Lock (Boston Scientific FloSwitch HP).
- Needle for initial arterial access (Boston Scientific Arterial Entry Needle 18 gauge, 2 3/4in.
- Torque device ( Terumo Torque Device).
- Perclose ProGlide Suture-Mediated Closure Device (Abbott Vascular, for 5-8F access sites) or Prostar XL Suture-Mediated Closure Device (Abbott Vascular, 6.5-10F access sites).
Tips & Pitfalls
- Cannulation of the contralateral limb gate is the portion of the operation that requires the most “wire skills”. This portion may become lengthy and discouraging if the surgeon fails to cannulate the gate using a particular approach. The authors recommend setting a specific time period for retrograde cannulation of the contralateral gate, and to proceed to the next step (antegrade or crossover cannulation) once this time has elapsed. The brachial/artery approach is the next step in the algorithm.
- The skills necessary to perform EVAR can not be attained through short “courses.” A dedicated formal training (six months minimum!) is necessary in order to obtain the “wire skills” and to independently perform these endovascular procedures. The trained endovascular surgeon should be able to deal with intraoperative complications of EVAR. Peripheral artery angioplasty/stenting should be part of the armamentarium of the endovascular surgeon. The current privileging criteria in most tertiary hospitals require a minimum of 100 diagnostic, 50 interventional and 10 EVAR cases to become certified/privileged.
- Most cardiothoracic surgeons will not meet the above training criteria (see also the credentialing section in the Endoluminal Technology Portal). In these cases, the necessary “wire skills” may be acquired by hiring a cardiothoracic surgeon who has had dedicated training in endovascular surgery. Alternatively, collaboration with other physicians with interventional skills (vascular surgeons, interventional radiologist/cardiologist) will be crucial. In addition to this, one may consider having a fellow surgeon to act as a mentor for the first five EVAR cases.
- Train a dedicated nursing team for your endovascular cases. An Endosuite will substantially improve your imaging capabilities and broaden your endovascular options.
- Remember the PPPPP rule: Prior planning prevents poor performance! Preoperative imaging and its accurate interpretation is essential in preoperative planning. Look for poor access vessels, i.e. significant calcification or tortuosity in the iliac arteries. It is important to have at least two backup plans, including open repair.
- The patient will need to be followed up very closely in the postoperative period. A contrast CT with delayed non-contrast scan is performed at one month, three months, six months, one year and annually thereafter to evaluate for endoleak, migration, device structural deterioration, etc. Alternatively, abdominal ultrasound and non-contract CT in adjunct with the CardioMEMS device (EndoSure Wireless AAA Pressure Measurement System; CardioMEMS, Inc.) may be used to monitor those patients with chronic renal failure. The close follow-up and the chance of postoperative issues requiring re-intervention must be discussed with the patient as part of the informed consent.
References
- Schneider PA. Endovascular Skills, 3rd Ed. New York: Informa 2009.
- Brewster DC, Cronenwett JL, Haller JW, et al. Guidelines for the treatment of abdominal aortic aneurysm: report of a subcommittee of the Joint Council of the American Association for Vascular Surgery and Society for Vascular Surgery. J Vasc Surg 2003;37:1106-1117.
- Greenhalgh RM, Brown LC, Kwong GP, et al. EVAR trial participants. Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysms (EVAR trial 1). Lancet 2004;364:843-848.
- Prinssen M, Verhoeven ELG, Buth J, et al. Dutch Randomized Endovascular Aneurysm Management (DREAM) Trial Group. A randomized trial comparing conventional and endovascular repair of abdominal aortic aneurysms. N Eng J Med 2004;351:1607-1618.
- Zarins CK, Crabtree E, et al. Endovascular aneurysm repair at 5 years: Does aneurysm diameter predict outcome? J Vasc Surg 2006;44:920-29.
- Lee WA, et al. Total percutaneous access for endovascular aortic aneurysm repair (“Preclose” technique). J Vasc Surg 2007;45:1095-101.
- Manual of Peripheral Vascular Intervention. Casserly IP, et al. Philadelphia: Lippincott Williams and Wilkins; 2005.
- Mastery of Vascular and Endovascular Surgery. Zelenock GB, et al.; Philadelphia: Lippincott Williams and Wilkins; 2006.