ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Surgery of the Superior Vena Cava: Resection and Reconstruction
Patient Selection
Resection and reconstruction of the superior vena cava (SVC) is still considered a surgical challenge; the numerous problems related to patient selection, choice of the appropriate surgical strategy and technique, the most suitable material for replacement, and the number of potential complications definitely show an impact on prognosis. However, with the appropriate indications and surgical technique a clear benefit has been documented in a selected group of patients[1, 2].
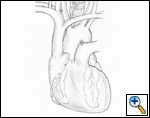 |
| Figure 1. Schematic anatomy of the superior vena cava. |
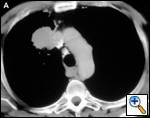
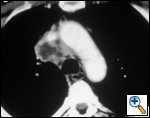
The anatomy of the SVC (Figure 1) and left brachiocephalic vein put this venous system in a critical area vulnerable to tumors arising both in the lung and anterior mediastinum. Malignant invasion is the most frequent indication for SVC resection and reconstruction. Lung cancer can involve the vessel with direct invasion by primary tumors arising in the right upper lobe or by nodal metastases (stations R2, R4 and 3) (Figure 2). Anterior mediastinal tumors (thymoma, thymic carcinoma, germ cell tumors, etc) may involve directly both the SVC and the left brachiocephalic vein (Figure 3). Primary tumors of the SVC represent a rare indication for surgery. Other infrequent indications are saccular aneurysms or primary malformations and traumatic lesions (iatrogenic, blunt, or penetrating injuries).
 |
 |
 |
| Figure 2A & B. (A) Lung cancer invading the superior vena cava. (B) Mediastinal lymph nodes invading the superior vena cava. | Figure 3. Cortical thymoma invading the superior vena cava. | |
Contraindications to surgical resection and replacement include the presence of SVC syndrome related to unresectable tumors, a completely obstructed SVC with a rich collateral vein circulation and abnormal walls of the proximal vein(s) (i.e., tumor involvement at the margins); particular attention needs to be directed to patients undergoing induction chemo-radiotherapy.
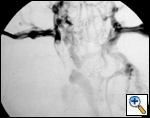 |
| Figure 4. Superior vena cavography showing obstruction by a mediastinal tumor. |
The preoperative work–up should always include total body CT scan for patients with lung cancer or tumors of the mediastinum. Other specific investigations should be tailored according to the histological diagnosis. Superior vena cavography should be performed when SVC invasion is suspected (Figure 4). Alternatively, magnetic resonance imaging can help to define the site and extension of the infiltration and the presence of thrombosis, and anatomical variations of the SVC system can be revealed. Echocardiography should be routinely included to rule out extension of a thrombus into the right atrium. Brain CT scan with contrast injection should always be performed for staging purposes (lung cancer) and also to rule out the presence of any brain disease that may be exacerbated by CNS edema during SVC clamping.
A careful functional evaluation should be performed (PFTs and arterial blood gas analysis) since a relevant number of patients with right upper lobe lung cancer invading the SVC are candidates for standard pneumonectomy or pneumonectomy with carinal resection.
Operative Steps
Surgical Approach
A right thoracotomy in the 4th or 5th intercostal space is the standard approach for upper lobe tumors invading the SVC. Complete median sternotomy is recommended for tumors of the anterior mediastinum. Both approaches allow optimal exposure of the operatory field; through the right thoracotomy we have easy access to the lung parenchima, the SVC, the trachea, the pulmonary hilum and right atrium; however, through this approach it is more difficult to control the left brachiocephalic vein. Median sternotomy allows a wide exposure of the mediastinum and the dissection can be easily extended into the neck. This approach can be turned into a “trap door incision” if a more comfortable exposure of the right lung and subclavian vessels is required.
Intraoperative management: Resection and reconstruction of the SVC is considered a major technical challenge due to the potential detrimental effects of clamping a patent vessel[3]. Partial caval clamping or clamping a chronically obstructed SVC is generally well tolerated; on the other hand, occlusion of a patent SVC may produce intracranial bleeding, brain edema and damage, and a potentially lethal reduction of cardiac output. These complications can be avoided by careful patient selection and intraoperative monitoring and management.
A double lumen endobronchial tube is placed to achieve one–lung ventilation. A radial arterial line and a venous line in the internal jugular vein are inserted routinely to obtain continuous pressure monitoring. Two additional venous lines are placed in the lower limbs to achieve volume expansion during venous clamping.
A Foley catheter is inserted to monitor urine output. EKG monitoring is obviously mandatory. Transesophageal echo is optional as well as nasogastric tube placement; the latter may be of help as an anatomical marker during dissection for lung cancer extensively involving the mediastinum.
Closure of up to 50% of the SVC circumference can be performed without any hemodynamic imbalance. In patients with tight or complete obstruction of the vessel venous clamping does not significantly modify cerebral circulation and cardiac output. In both cases the duration of venous clamping is not a limiting factor and the operation can be safely performed without feeling stressed by the potential length of the reconstructive step. During complete clamping of the unobstructed SVC, there is a clear hemodynamic derangement with an increase of the mean venous pressure in the cephalic districts and a decrease in mean arterial pressure with a consequent reduced brain arterial–venous gradient. This may lead to brain edema, hemorrhage and dysfunction (usually transient). These hemodynamic modifications are evident in the entire cephalic distribution. For this reason, a cyanotic facies is almost always present during SVC clamping; however, it is usually completely reversible after declamping. Petechiae may be evident in the immediate postoperative period and disappear within a couple of weeks after surgery.
The hemodynamic imbalance is reduced with an aggressive intraoperative management by the anesthetist, along with some technical tricks.
- Fluid implementation and pharmacological agents: this strategy is devoted to increase venous return and maintain the normal arterial–venous gradient in the brain. Macromolecules, blood and plasma should be used. Vasoconstrictive agents are indicated to increase the mean arterial pressure. At the end of the procedure diuretics are administered to reduce edema in the cephalic regions. Anticoagulation therapy is mandatory during and immediately after the operation: intravenous sodium heparin (0.5 mg/Kg) is given before clamping and continued during the immediate postoperative period (adjust heparin dose to achieve INR = 2 to 2.5). It is usually switched to warfarin agents at the time of discharge.
-
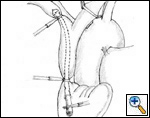
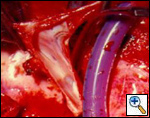
Figure 5A & B. (A) Intraluminal shunt. (B) Extraluminal shunt during reconstruction of the superior vena cava. Surgical strategy and shunting techniques: Every effort should be attempted to reduce clamping time as much as possible, in particular when the SVC system is not completely occluded before the operation. It has been reported that up to 45 to 60 minutes of complete clamping is usually tolerated with the appropriate pharmacological support. For lung cancer resection, the vascular step should be always performed before any other reconstructive procedure of the airway. For mediastinal tumors involving the upper lobes, the dissection should be performed from the left to the right side; the right part of the excision is usually performed after vascular reconstruction, in particular when a lobectomy is required. Intravascular or extravascular shunts (Figure 5) may be used to reduce the effects of vascular clamping during resection and reconstruction of the SVC[4]. However, thrombosis of the shunt may occur; furthermore, these devices occupy space in the operative field making the anastomosis more difficult.
 |
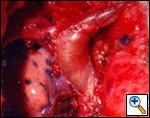 |
| Figure 6. Tangential resection of the superior vena cava with direct suture. | Figure 7. Reconstruction of the superior vena cava and left brachiocephalic vein with a patch of autologous pericardium. |
Surgical Technique
Tangential resection and venous plasty: in cases in which less than 30% of the SVC circumference is involved, a partial resection of the vessel can be performed. For minimal invasion, a tangential resection of the defect closed with a running suture or a vascular stapler is usually easy and leaves a patent vessel (Figure 6). Larger defects, especially if they are longitudinally extensive, require reconstruction; this can be obtained by the interposition of a patch of autologous or bovine pericardium (Figure 7). Autologous pericardium may be fixed in diluted glutaraldehyde (two drops of 20% glutaraldehyde in 50 cc of saline) for one minute to let it stiffen and facilitate manipulation during suturing.
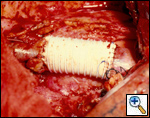
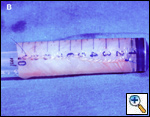
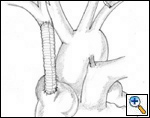
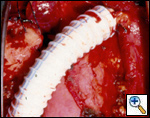
SVC replacement: replacement of the whole body of the SVC is the most frequent type of reconstruction (Figure 8). In patients with lung cancer, it is usually associated with right upper lobectomy or pneumonectomy; however, SVC reconstruction may be required during carinal pneumonectomy, sleeve lobectomy (Figure 9), and reconstructive procedures of the pulmonary artery[5]; the latter two should always be attempted if they help to avoid pneumonectomy. SVC replacement requires a tumor–free confluence of both brachiocephalic veins. The reconstruction is usually performed using a straight non-ringed PTFE graft (18 – 20 mm). An autologous or bovine pericardial tube could also be used (Figure 10). After proximal and distal clamping, the SVC is excised. The proximal anastomosis is performed first, using a 5-0 polypropylene suture, starting from the posterior aspect of the prosthesis or the tube of pericardium. The distal anastomosis is subsequently performed with the same technique. Before tying the distal suture line, the proximal clamp is gently released and deaeration is performed; afterwards, the distal clamp is released and knots are tied. After complete filling of the graft by blood, there should be no tension of the suture lines or torsion or kinking.
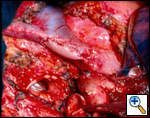
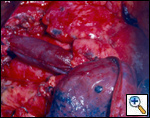
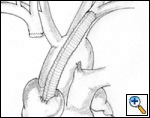
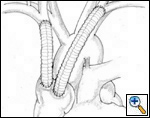
Sometimes it may be indicated to replace only one brachiocephalic vein (either the right or left, according to local invasion). A ringed PTFE graft should be used (Figure 11, 12), especially for the left vessel and the distal anastomosis should be performed on the inferior stump of the SVC or on the right atrium. Use of the auricle should be avoided because of the presence of the pectinate muscles. In this situation, the prosthesis may be too long after closure of median sternotomy and could kink easily. Also postoperative mediastinal fibrosis (especially if radiotherapy is administered) could contribute to graft compression. Simultaneous revascularization of both brachiocephalic veins (Figure 13) is rarely required since there are enough anastomotic venous communications in the neck. Revascularization of both systems is indicated in case of previous neck surgery (laryngeal or thyroid surgery for cancer); a separate distal anastomosis of the two grafts is preferred to avoid thrombosis of the accessory left limb of a Y graft, starting at the level of the graft–to–graft anastomosis.
Palliative bypass: the indications for palliative procedures of bypass are extremely rare due to the low venous blood flow obtained from the axillary or jugular veins. Surgically created A–V fistulae devoted to increase flow through the bypass conduit are generally unsuccessful.
Preference Card
- 2.5 magnification loupes
- Headlight
- Long (24 cm) heavy titanium Castro–Viejo needle holder when performing the vascular sutures through lateral thoracotomy; standard Castro–Viejo needle holder when working through median sternotomy.
- Straight ringed and unringed PTFE grafts (from 12 mm to 20 mm)
- Bovine pericardium
- Glutaraldehyde (two drops of 20% glutaraldehyde in 50 ml of saline)
- Vascular shunts
Tips & Pitfalls
- Carefully select candidates for SVC resection and reconstruction.
- Never deny this procedure to patients who correctly fit all the indications, but don’t hesitate to avoid surgery when contraindications are present. Don’t be attracted by the chance to perform a technically demanding operation.
- Patients with primary tumors of the mediastinum are excellent candidates; after radical resection, long-term survival is extremely encouraging.
- Patients with N2 lung cancer are not ideal candidates from the oncological point of view; no long-term survivors have been reported. Also, incomplete resection is considered a negative prognostic factor.
- Avoid SVC resection and revascularization in patients with excellent collateralization towards the inferior vena cava.
- Do not dissect too much of the SVC and the two brachiocephalic veins on the side of the proximal anastomosis; you need just enough space to clamp the vessel and perform the anastomosis. Excessive mobilization could favor twisting and stenosis of the proximal suture line.
- Choose the appropriate graft size to allow optimal flow (18 mm to 20 mm for replacement of the body of the SVC, 12 mm to 13 mm for the left brachiocephalic vein, 12 mm to 14 mm for the right brachiocephalic vein).
- Choose the appropriate length of the graft to avoid kinking, especially when replacement of the left brachiocephalic vein is performed through a median sternotomy.
- Heparinization is mandatory as in every vascular reconstruction.
- When reconstructive surgery of the airway is part of the operative procedure, always avoid contamination of the vascular surgical field. The SVC and tracheal suture lines should be separated by interposing a pedicled graft (intercostal muscle).
Results
A number of potential complications may be associated with resection and reconstruction of the SVC. Anastomotic stenosis may occur both at the level of the proximal and distal suture lines. Early diagnosis is extremely important; for this reason, a postoperative angiogram or MR should always be performed before discharge. Proximal kinking is the most frequent cause of stenosis; this may be due to excessive dissection and mobilization, especially if the left brachiocephalic vein is reconstructed through a median sternotomy. Also, excessive length of the graft may cause kinking. Management of this complication includes surgical correction in the early postoperative period and dilation and stenting when it is diagnosed later.
Other potential complications are graft thrombosis and infection. The former usually occurs early after the operation and may cause acute SVC syndrome. Graft infection is a serious complication and occurs more frequently when the vascular reconstruction is associated with major airway surgery (tracheal or bronchial sleeve resections). Conservative management of this complication is possible when the patient is not septic. In case of septicemia, the graft should be removed, possibly after progressive clamping of the prosthesis through an open window, as described by other authors[6].
Operative mortality should be between 5% and 10%. The survival rate after radical resection of mediastinal tumors invading the SVC is excellent: 60% at 5 years according to Dartevelle and colleagues[1]. Patients with lung cancer show a less favorable prognosis: about 30% at five years. There are no long-term survivors among patients with N2 disease; the potential role of induction chemotherapy in this group of patients is still to be defined[7].
References
- Dartevelle PG, Chapelier AR, Pastorino U, et al. Long-term follow–up after prosthetic replacement of the superior vena cava combined with resection of mediastinal–pulmonary malignant tumors. J Thorac Cardiovasc Surg 1991;102:259–265.
- Tsuchiya R, Asamura H, Kondo H, Goya T, Naruke T. Extended resection of the left atrium, great vessels, or both for lung cancer. Ann Thorac Surg 1994;57:960–965.
- Gonzalez–Fajardo JA, Garcia–Yuste M, Florez S, Ramos G, Alvarez T, Coca JM. Hemodynamic and cerebral repercussions arising from surgical interruption of the superior vena cava. Experimental model. J Thorac Cardiovasc Surg 1994;107:1044–1049.
- Piccione Jr,W, FaberLP, Warren WH. Superior vena caval reconstruction using autologous pericardium. Ann Thorac Surg 1990;50:417–419.
- Solli P, Spaggiari L, Grasso F, Pastorino U. Double prosthetic replacement of pulmonary artery and superior vena cava and sleeve lobectomy for lung cancer. Eur J Cardiothorac Surg 2001;20:1045–1048.
- Dartevelle P, Macchiarini P, Chapelier A. Technique of superior vena cava resection and reconstruction. Chest Surg Clin N Am 1995;5: 345–358.
- Rendina EA, Venuta F, De Giacomo T, et al. Induction chemotherapy for T4 centrally located non-small cell lung cancer. J Thorac Cardiovasc Surg 1999;117:225–233.




Comments